/PeriodicTableoftheElements-5c3648e546e0fb0001ba3a0a.jpg)
To confirm your answer, compare the calculated value to the weighted mass displayed on the periodic table. 
The atomic mass of oxygen (use percent natural abundance data from Table 2.5.1) would be calculated as follows:Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + (%3)(mass 3) Therefore, the molar mass of a substance calculated by adding the atomic masses of each element present in that molecule. This looks a lot like a conversion factor that relates two units: grams to moles. When the chemical elements are thus arranged, there is a recurring pattern called the periodic law in their properties, in which elements in the same column (group. For chlorine, there are only two naturally occurring isotopes so there are only two terms.Ītomic mass = (%1)(mass 1) + (%2)(mass 2) + ⋯Īnother example: oxygen exists as a mixture that is 99.759% 16O, 0.037% 17O and 0.204% 18O. Notice that the units on molar mass are grams per mole (g/mol). periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number i.e., the total number of protons in the atomic nucleus. We know that mass increases as the atomic number. This is repeated until there is a term for each isotope. Weve discussed mass at length by doing molar mass conversions. The weighted average is determined by multiplying the percent of natural abundance by the actual mass of the isotope. Relative atomic mass is the ratio of the average mass of the atom to the unified atomic mass unit (symbol: u) or dalton (symbol: Da), which is defined as one twelfth of the mass of a carbon-12 atom in its ground state. The atomic mass of an element is the weighted average of the atomic masses of the naturally occurring isotopes of that element. Atomic Mass in the Periodic Table of Elements. The numbers in the periodic table that we identified as. 
Why? The reason is that we need to take into account the natural abundance percentages of each isotope in order to calculate what is called the weighted average. One mole of a substance has the same mass in grams that one atom or molecule has in atomic mass units. One mole of a substance has the same mass in. The lessons learned in this post will continue through the study of chemical reactions and equations.\( \newcommand = 35.968 \,u \nonumber \]Īs you can see, the average atomic mass given in the last column of the table above (35.453) is significantly lower. And whereas one sodium atom has an approximate mass of 23 u, 1 mol of Na atoms has an approximate mass of 23 grams. 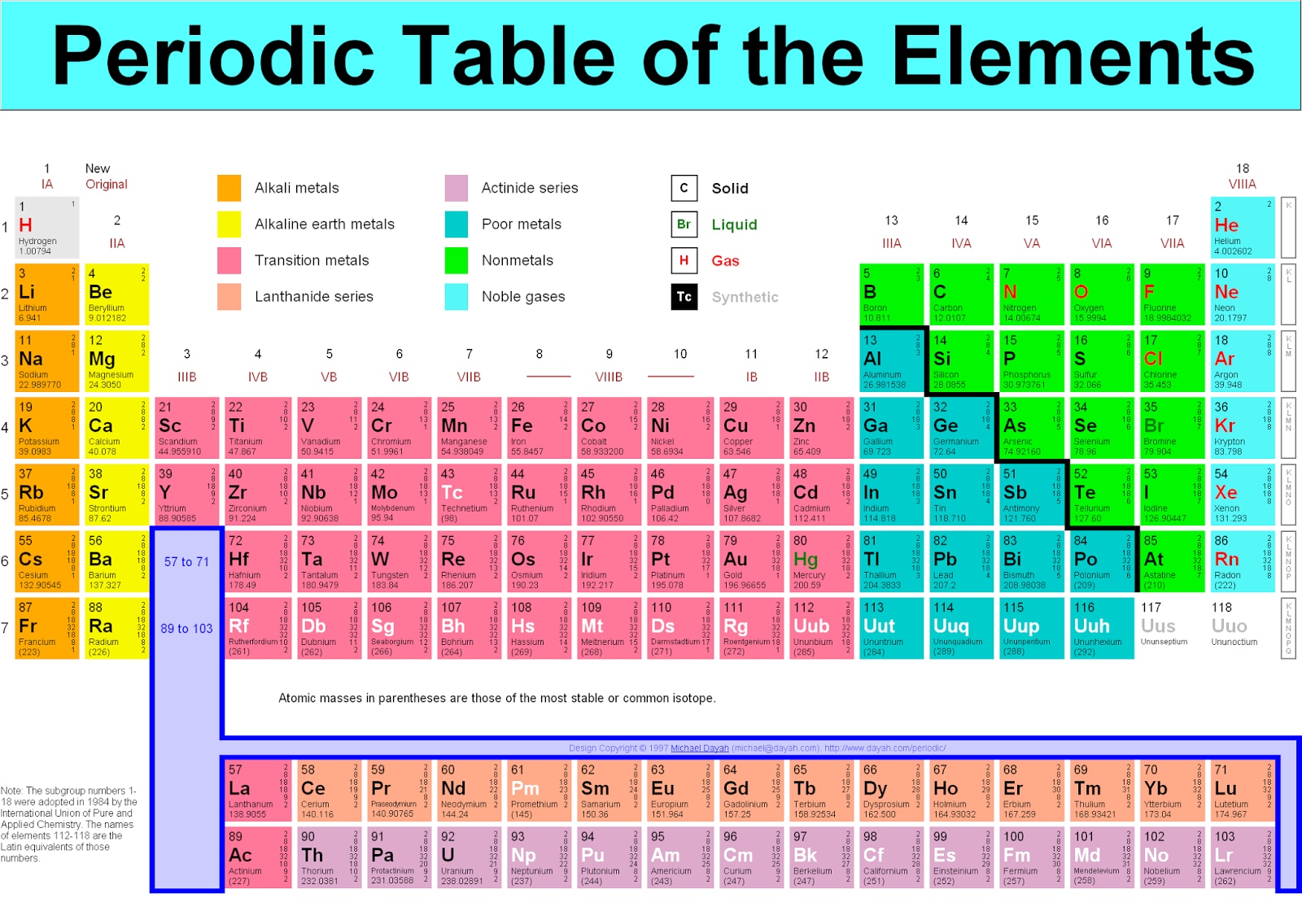
We also learned how to calculate the molar mass of a compound using the periodic table and how to convert mass into moles. You can also use this molarity calculator to find the mass concentration or molar mass. Substitute the known values to calculate the molarity: molarity 5 / (1.2 × 36.46) 0.114 mol/l 0.114 M. As mass / volume molarity × molar mass, then mass / (volume × molar mass) molarity. We learned about Avogadro’s number and how it relates to the mole, which is a unit used to express the amount of a substance. Convert the expressions above to obtain a molarity formula. Element Sulfur (S), Group 16, Atomic Number 16, p-block, Mass 32.06. In conclusion, understanding molar mass is an essential concept in chemistry as it allows us to relate the amount of a substance to the number of particles present in it. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Atomic Weight is the molar mass of an element. There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. Please note that the elements do not show their natural relation towards each other as in the Periodic system. It is known that the chlorine molecule is diatomic - Cl2, then the relative atomic mass of the chlorine molecule will be. The unity for atomic mass is gram per mol. Specifically, the number is defined as 6.022 \times 10^. The unit for molar mass (note it is the mass of a mole) is grams/mole. The lightest chemical element is Hydrogen and the heaviest is Hassium. Example 3: Finding the Mass of a Number of MoleculesĪvogadro’s number is a fundamental constant that represents the number of particles (atoms, molecules, ions) in one mole of a substance.How to Find Molar Mass Using the Periodic Table.The Mole and its Relationship to Avogadro’s Number.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
